|
And if we look at our rules, we see that sodium bromide is soluble and magnesium sulfide is soluble because it is one of the exceptions. Example 4: Take two aqueous solutions of cadmium sulphate (CdSO 4) and potassium sulphide (K 2 S). 
The balanced chemical equation is the net reaction for formation of Fe (OH)3 Fe (ag) + 3 OH (a g) Fe (OH)3 (. The ionic equation for the precipitation reaction of aqueous NaOH and aqueous MgCl 2 is: N a+OH +M g+ 2Cl M g+ 2(OH) 2 +2N a+Cl N a + O H + M g 2 + C l M g 2 + ( O H) 2 + 2 N a + C l The precipitate of Mg (OH) 2 is white in color. 
We need a balanced chemical equation and amounts in moles to calculate how much precipitate forms. So we are going to put a two in front of our sodium bromide and that balances it. For example, a precipitate of Fe (OH) forms when 50.0 mL of 1.50 M NaOH is mixed with 35.0 mL of 1.00 M FeCl3 solution. If we can see that we have to sodium is on the left and only one on the right. A precipitation reaction is a double-displacement reaction because it takes place between two ionic compounds which form an insoluble product. So now for our balanced equation we have sodium sulfide plus magnesium bromide forms sodium bromide plus magnesium sulfide. And then we have magnesium And sulfur coming together and that's just plus two and -2. This is the net ionic equation because some of our ions arent taking part in this. We could show our silver cations and our chloride anions combining to form our precipitate, AgCl. We could show the silver cations here, Ag+. And we have our sodium and our bromide coming together and we're going to crisscross our charges and it is A plus one and a negative one. We could write a net ionic equation showing whats happening to form our precipitate. So we have sodium sulfide plus magnesium bromide. Um barium and magnesium and we have carbonates except when with group A. 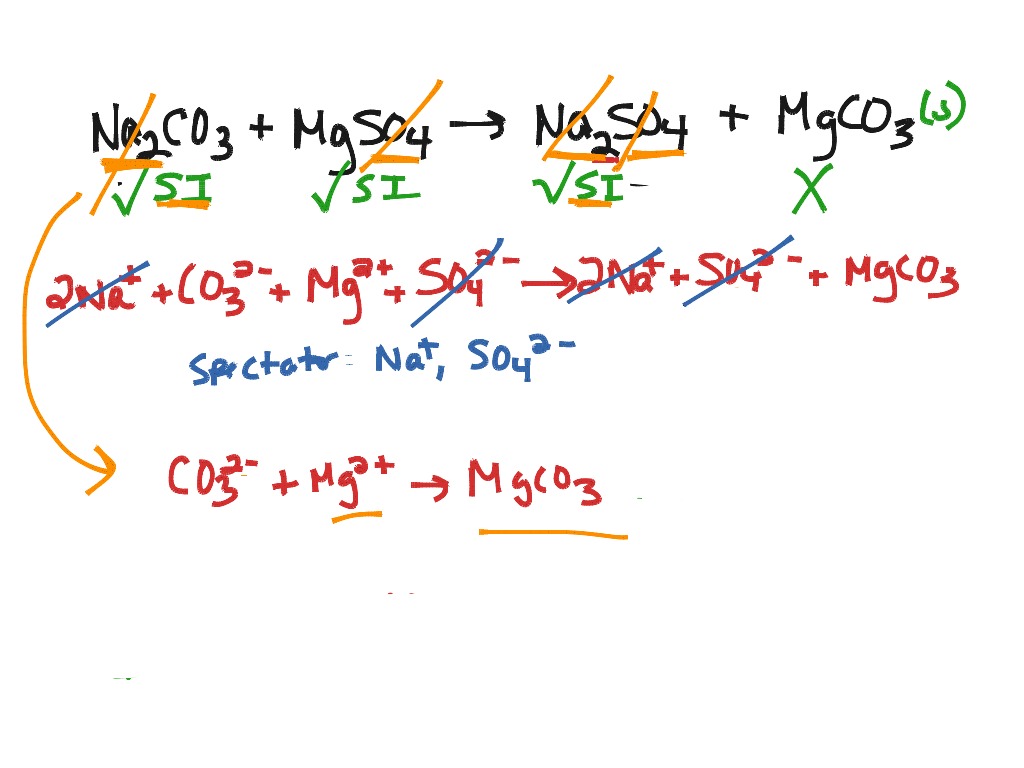
And now we have our insoluble als and that is hydroxide except when With Group one A. Complete ionic equation: All strong electrolytes written as dissociated ions weak electrolytes, precipitates, and gases are written as undissociated. And that is except when with silver would copper or mercury. And we have nitrates acetate and most her chlorides. So we have soluble and then we have some exceptions. So let's remember our soluble itty rules. Hi everyone here we have a question telling us to consider an aqueous solution containing sodium sulfide and magnesium bromide, identify the precipitate if any and write the balanced equation. The first step to writing a net ionic equation is to separate the soluble (aqueous) reactants and products into their respective cations and anions.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
